Oryzon Genomics (MADX: ORY) is a clinical stage biopharmaceutical company headquartered in Barcelona, Spain. In December 2015, I provided a detailed overview of the company for investors (download the 19-page report). Oryzon is a leader in the development of epigenetics-based therapeutics. Epi is Greek for above, and genetics is the study of genes, heredity, and genetic variation in living organisms. As such, epigenetics is the study of “above the genes”, or more specifically, modifications that occur to the genes that result in activation or deactivation of gene expression without alteration in DNA sequence. These modifications are known as epigenetic modifications. The company is applying epigenetics to drug discovery and development in the area of cancer and neuroinflammatory / neurodegenerative diseases.
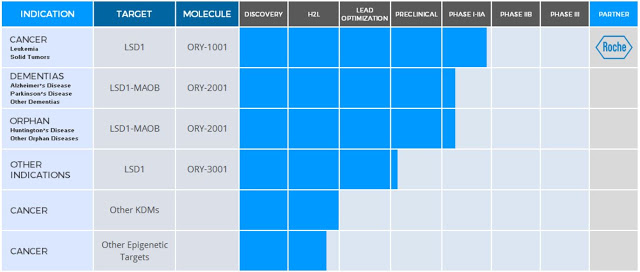
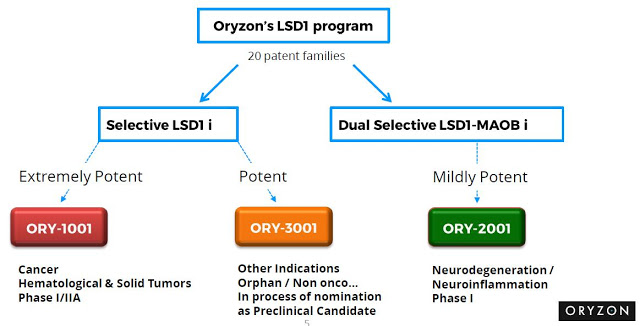
Oryzon has made significant progress with its development programs over the past year. The company’s lead candidate, ORY-1001, has progressed into the second part of a Phase 1/2a clinical study. ORY-1001 is being developed in collaboration with Roche for hematological and solid tumors. Management expects to report preliminary data from this program at the American Society of Hematology (ASH) meeting in December 2016. A second candidate, ORY-2001, has entered the clinical for the treatment of Alzheimer’s disease. Oryzon is currently conducting a Phase 1 multiple ascending dose study with ORY-2001 in healthy volunteers. A Phase 2 study is expected to start during the first half of 2017. Recently, a third candidate, ORY-3001, has been nominated for advanced preclinical testing; a Phase 1/2a study is planned for the second half of 2017.
Below is an update on Oryzon Genomics following the company’s second quarter 2016 financial results.
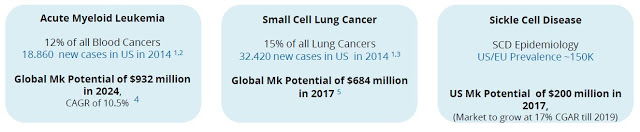
Oryzon Genomics lead clinical-stage candidate is ORY-1001, a potent and highly selective LSD1 inhibitor. LSD1 (also known as KDM1A) plays a key role in the regulation of gene expression and regulation of several proliferation-associated genes implicated in aggressive cancer biology. Preclinical work with ORY-1001 has demonstrated highly (subnanomolar) specific and potent activity against LSD1. Pharmacokinetic data suggests good oral bioavailability with druggable ADMET (absorption, distribution, metabolism, excretion, and toxicity) properties. The company is focusing initial development of ORY-1001 on myeloid malignancy, a pathology in which epigenetic dysfunction plays a central role. Proof-of-concept for this approach has been demonstrated in a mouse model of human MLL-AF9 leukemia. ORY-1001 was granted Orphan Drug designation by the European Medicines Agency for Acute Myeloid Leukemia (AML) in July 2013.
In April 2014, Oryzon announced they have entered into a worldwide collaboration to research, develop and commercialize inhibitors of LSD1 (KDM1A), including lead molecule ORY-1001, with Roche. Under terms of the agreement, Roche will have sole responsibility for developing and commercializing ORY-1001 and/or its backup compounds following completion of the ongoing Phase 1/2a clinical trial. The agreement includes the licensing of two patent families that Oryzon has created around LSD1, and includes options for other Oryzon programs to be incorporated in future.
In return for licensing these rights to Roche, Oryzon received an upfront payment and milestones totaling $23 million to date, plus potential development, commercial and sales milestone payments across hematology, cancer, and non-malignant indications that could exceed $500 million, together with tiered royalties on sales which range up to mid-double digits. In May 2016, the R&D collaboration with Roche to explore oncology and hematology indications for ORY-1001 was extended by Roche until March 2017. The extension allows for Oryzon to gain additional insight into the drug’s mechanism of action, provided support for its use in indications beyond acute leukemia, and expand the toolbox for future and current clinical trials. The main goal over the next several months is to finalize the transfer of the newly generated technology and knowledge over to Roche.

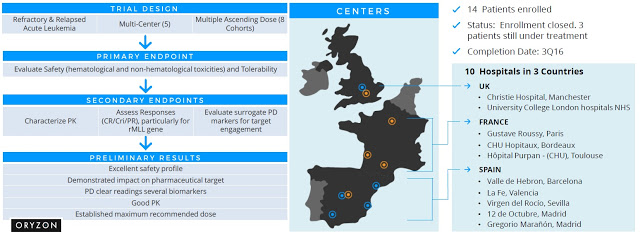
Oryzon is currently conducting a Phase 1/2a clinical study with ORY-1001 in patients with refractory or relapsed Acute Leukemia. The Phase 1 portion of the trial was a multicenter, multiple ascending dose escalation study designed to ensure the safety (hematological and non-hematological toxicities), tolerability, and pharmacokinetics of ORY-1001 in patients with refractory and relapsed acute leukemia. This was successfully completed in late 2015. Preliminary results obtained from the trial demonstrate excellent safety, pharmacokinetics and pharmacodynamic measures, as well as target biology. No serious drug-related adverse events have been reported, and the company believes they have established the maximum recommended dose for future clinical studies. This work was done at five clinical sites, four in Spain and one in the UK.
Oryzon has since expanded this study into five additional centers in France, Spain, and the UK for the Phase 2a portion of the trial. The enrollment criteria have also been focused to include patients with target mutations such as mixed-lineage leukemia (MLL) and M6. This is a rare subset of disease in which leukemia stem cells are especially sensitive to LSD1 inhibition. The expansion will allow the company to obtain preliminary signs of efficacy for ORY-1001, setting the stage for future development work to be conducted by Roche. The first patient in the Phase 2 portion of the trial was enrolled in November 2015. Preliminary data are expected to be reported at the American Society of Hematology (ASH) meeting in late 2016.
Beyond AML, Oryzon believes that ORY-1001 has utility in solid tumors. For example, LSD1 is overexpressed in multiple tumor types and the ORY-1001 mechanism of action suggests anti-tumor beyond leukemia. GlaxoSmithKline has progressed into Phase 1 clinical development with a similar LSD1 inhibitor, GSK2879552, for small cell lung cancer (SCLC), and based on published data; ORY-1001 looks several times more potent. As such, Roche is expected to follow Oryzon’s initial work in AML with a clinical program in SCLC in 2017.
Beyond oncology indications, the potential for LSD1 inhibitors in non-oncology hematological indications is being explored, for example in sickle cell disease (SCD), a rare genetic disorder affecting an estimated 70,000 to 100,000 individuals in the U.S. Independent data out of the University of Chicago on LSD1 inhibition was recently disclosed at the 56th American Society of Hematology (ASH) annual meeting in December 2014 using an Oryzon tool compound. The data highlight the potential for LSD1 inhibitors as treatment options for SCD.
In total, ORY-1001 is targeting potential indications with sales in the billions of dollars. With Roche taking over development responsibilities for the clinical development of ORY-1001 following the completion of the current Phase 2a trial, it will be exciting to see where they take the drug in 2017.
Update On ORY-2001
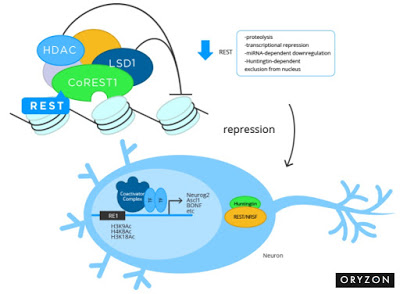
Oryzon’s second drug candidate is ORY-2001, a potent and selective, orally available, small molecule inhibitor of LSD1-monoamine oxidase-B (MAO-B). Epigenetic inhibition of LSD1-MAO-B has been shown to have a downstream effect on numerous genes and different biological pathways resulting in consequent transcriptional dysregulation. LSD1 is a key component of the LSD1-REST- CoREST-HDAC1/2 repressor complex involved mainly in controlling developmental programs and modulating neuronal morphology in the CNS. LSD1 is also known to be an important regulator in the maintenance of pluripotency and in specification of neuronal commitment of pluri- or multipotent cells. This mechanism is believed to be an important marker of disease status and its progression in many neurodegenerative diseases, such as Alzheimer’s disease.
Initial pharmacology data with ORY-2001 suggests the drug has an excellent pharmacokinetic profile, with a wide therapeutic window and convenient once-daily dosing. Data shows the drug crosses the blood-brain barrier with highly selective anti-LSD1 and MAO-B activity, with no off-target binding.
Preclinical data suggest that ORY-2001 improves cognition, with positive implications for diseases such as Alzheimer’s and Huntington’s disease. For example, in a non-transgenic mouse model of AD, company scientists saw a marked cognitive improvement correlating with changes in the expression of key genes in the hippocampus. In studies partially supported by the Alzheimer’s Drug Discovery Foundation, company scientists showed that ORY‐2001 provided a dose‐responsive protective effect in the medium and long‐memory of female mice, compared to age-matched SAMP8 mice (see below). Additional preclinical studies with ORY-2001 show improvement of survival and recovery of phenotypic characteristics in mouse models of HD and other neurodegenerative disorders.
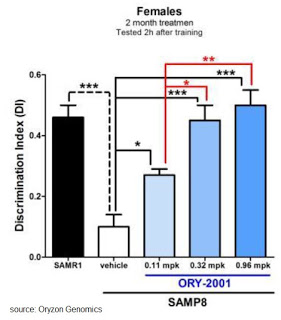
Oryzon has identified different hippocampal biomarkers relative to ORY-2001 treatment. ORY-2001 potently down-regulates the expression of a subset of genes related to immune reactions and inflammation, including S100A9 and T-cell receptor b chains in SAMP-8 mice. Down-regulation of the pro-inflammatory S100A9 protein by ORY-2001 is particularly interesting since S100A9 is emerging as an important contributor to inflammation-related neurodegeneration. For example, S100A9 was found to be increased in patients with Alzheimer’s disease, post-operative cognitive dysfunction, and traumatic brain injury (see work by Wang C. et al., 2014). Knockout or knockdown of S100A9 has been shown to be beneficial to memory in APP/PS1 and Tg2576 models of Alzheimer’s disease. Additionally, preclinical data shows that ORY-2001 up-regulates genes associated with improved cognitive function, neuroplasticity, and memory.
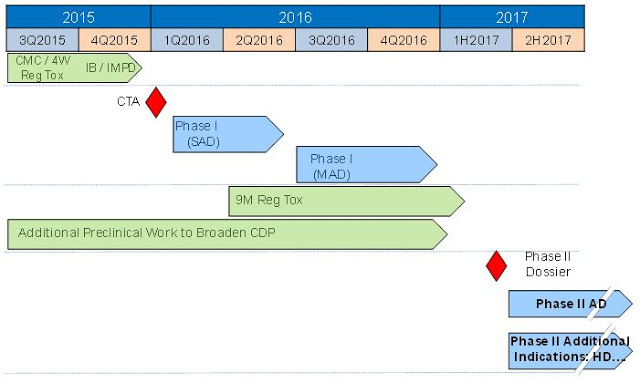
Oryzon received approval in Spain to initiate a Phase 1 study for ORY-2001 in January 2016. The randomized, double-blind, placebo-controlled single and multiple ascending dose program is designed to investigate the safety, pharmacokinetics, and pharmacodynamics of oral ORY-2001 in healthy subjects as well as an elderly population. The trial is being conducted at a university hospital in Barcelona, Spain. The first patient was dosed in April 2016. The multiple ascending dose cohort began enrolling patients in July 2016. Target enrollment is 48 subjects. I expect that with positive data, Oryzon will move ORY-2001 into a Phase 2 study in patients with Alzheimer’s disease during the first half of 2017. A timeline for future development or ORY-2001 pulled from the company’s most recent investor presentation can be seen below.
Oryzon owns full rights to the drug; thus, the Phase 2 study could represent a significant valuation inflection for the company if successful. Alzheimer’s disease is obviously an enormous market, with approximately 5.4 million Americans affected today. The number is expected to rise to 7.1 million in the U.S. by 2015 according to Alz.org. Alzheimer Europe estimates 8.7 million Europeans are affected by Alzheimer’s disease. Another 10 to 12 million people in Asia are also suspected to suffer from Alzheimer’s. Little success has been accomplished with respect to new Alzheimer’s treatments over the past decade. An epigenomic approach to Alzheimer’s, as well as Huntington’s disease and Parkinson’s disease, represents a novel and exciting new approach to these difficult to treat diseases.

Update On ORY-3001
In July 2016, Oryzon announced that it had designated its next drug, ORY-3001, for preclinical development. ORY-3001 is a first-in-class specific Lysine Specific Demethylase 1 (LSD1) inhibitor for the treatment of, yet undisclosed, non-oncological conditions. ORY-3001 is an enantiomerically pure, potent and selective compound with good pharmacological properties, orally bioavailable, with optimal PK, safety and selectivity profile. After successful completion of regulatory toxicology studies, the company expects to move ORY-3001 into clinical Phase 1/2a studies during the second half of 2017.
Update On The Financial Position
On August 8, 2016, Oryzon reported financial results for the first half ending June 30, 2016. Revenues totaled $2.7 million during the first six months of the year compared to $5.2 million in the first half of 2015. The loss for the quarter totaled $3.1 million. R&D investments are $2.5 million. Oryzon continues to invest in its therapeutic pipeline, having recently advanced ORY-2001 into a multiple ascending dose study and nominated ORY-3001 for IND-enabling studies in non-oncological conditions.
Oryzon ended June 2016 with $33 million in cash, equivalents, and short-term investment. Cash increased by $25 million from June 2015 thanks to financial loans, grants, collaborative research programs, and a recent debt financing. For example, during 2015, the company received a total of €5 million from its collaborative research program with Roche and €2.6 million from non-dilutive grants that support work on ORY-2001.
In May 2016, Oryzon closed on a debt financing securing €10.5 million ($12.1 million) in cash. In total, Oryzon has raised more than €27 million in cash since July 2015. Total financial debt stands at €23 million. The current cash and short-term financial investments are €33 million. I project operating burn over the next year will be between approximately €12-14 million. Thus the current balance is sufficient to fund operations for the next 18 months. There are approximately 28.5 million shares outstanding.
Valuation
Oryzon Genomics currently trades on the Madrid Stock Exchange (MADX: ORY) with a market capitalization of approximately €84 million. In my initial article back in December 2015, I outlined for investors why I believe the company is worth €200 million ($225 million USD) in value. I used a combination of peer-valuation, looking at recent deals between large pharmaceutical companies and smaller players in the epigenomic space, including the recent takeover of Tensha, a single asset spin-off company from Dana Farber that had a Bromodomain inhibitor in Phase 1b and was acquired by Roche for $115 million upfront in January 2016 plus contingent payments that may reach up to $420 million, the Merck-OncoEthix deal valued at $110 million upfront plus $155 million in contingent payments, the Celgene-Quanticel deal valued at $100 million plus up to $285 million in contingent payments, and the Gilead-EpiTherapeutics deal valued at $65 million, and discounted cash flow based on the forecasted commercial success of ORY-1001 and ORY-2001.
Since that time the company has made significant progress with the advancement of its pipeline. ORY-1001 is progressing in a Phase 2 clinical study, and preliminary results are expected at the ASH conference in December 2016. Peer-valuation analysis suggests a Phase 2 asset in oncology is worth approximately $150 million (see below). However, positive data at ASH and the fact that the company has partnered with Roche and could receive up to $500 million in milestones on ORY-1001 has the potential to create dramatic upside to this estimate. The ORY-1001 presentation at ASH should act as a major catalyst for the shares heading into 2017.
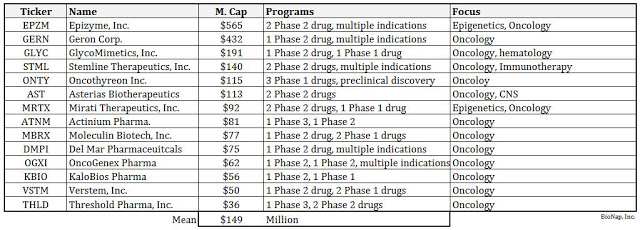
Oryzon has advanced ORY-2001 into a Phase 1 multiple ascending dose study. I expect that with positive data, Oryzon will move ORY-2001 into a Phase 2 study in patients with Alzheimer’s disease during the first half of 2017. Recently positive preclinical data with ORY-2001 sets the stage for a potential expansion into other types of dementia. Peer-valuation analysis suggests a Phase 2 Alzheimer’s / dementia asset is worth approximately $100 million in value.

Commensurate with the company advancing ORY-2001 into Phase 1 trials and Roche extending the R&D collaboration to explore additional oncology and hematology indications for ORY-1001, we now believe Oryzon Genomics is worth €225 million ($250 million USD) in value. We have yet to factor in any valuation for ORY-3001.
– Discounted Cash Flow
ORY-1001 is currently in a Phase 2 clinical study. Management expects the drug to be in Phase 3 in 2018. This suggests the NDA will be filed by Roche in 2020, putting the company in a position to gain approval for ORY-1001 in 2021. Oryzon is entitled to royalties and milestones potentially worth up to $500 million in value. The exact terms of the payments and royalty schedule have not been made public by Oryzon, but a model can be built nevertheless using best-guess assumptions. Assumptions for the success of follow-on indications in small-cell lung carcinoma and non-oncology uses can also be made.
ORY-2001 is currently in a Phase 1 multiple ascending dose study. Developing drugs for neurodegenerative diseases such as Alzheimer’s and Huntington’s disease tends to take longer than with terminal cancers such as AML. Accordingly, the NDA on ORY-2001 in the first indication, assuming that is Alzheimer’s disease, is not likely to be filed before 2022. Best-guess assumptions for success in Huntington’s can also be made, with appropriate discount and probability adjustment.
Below is a model showing the hypothetical ramp of ORY-1001 and ORY-2001. An appropriate “probability adjustment” to account for the early-stage nature of these programs has been applied to these forecasts. The model shows fair-value at $250 million, consistent with the peer-valuation analysis presented above.
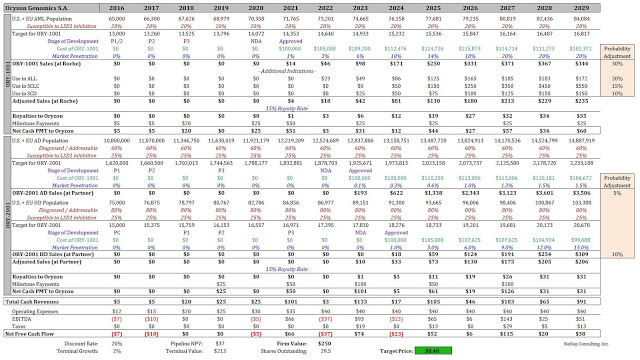
Oryzon is currently trading at €2.90 per share on the Madrid exchange. Our price target is $8.50 USD, or €7.75 per share for MADX:ORY.
Conclusion
Young, healthy cells have an epigenetic setting that promotes the formation of repressive heterochromatin while allowing expression of housekeeping genes and genes involved in cell cycle control, stress resistance, and DNA repair. During aging, cells acquire epigenetic modifications, also known as methylation drift, which facilitates activation of undesirable chromosomal regions and repression of cell cycle and control genes. Some of these regulatory and control genes include very important tumor suppressor genes, while others might include genes that are involved in cell differentiation or specification, or play a role in protein folding and aggregation. Cell stress and methylation drift have been shown to contribute to the aging phenotype; however, these modifications are also susceptible to epigenetic alterations through pharmacology. And, since epigenetic alterations are more readily reversible than genetic alterations, interventions aimed to reverse undesirable epigenetic changes may have great potential to treat age-associated diseases, including various cancers and neurodegenerative disorders (Muñoz-Najar & Sedivy, 2011).
Oryzon is focused on developing epigenetic-based therapies and personalized drugs from its proprietary platform technology. The pipeline includes one compound in Phase 1/2a in oncology, ORY-1001, a highly potent LSD1 inhibitor with exquisite selectivity that has been granted orphan-drug status by the EMA for acute myeloid leukemia, a second compound in Phase 1 clinical trials, ORY-2001, for the treatment of Alzheimer’s Disease, and additional programs in other cancer indications in various stages of preclinical development. Based on a review of available literature and analysis of peers, the company’s strategy seems sounds and likely to create significant shareholder value if successful.
We see fair value at €7.75 per share.


