Last week I wrote an article on Immune Pharmaceuticals (IMNP) and its first-in-class fully human IgG4 monoclonal antibody, bertilimumab, for the treatment of inflammatory bowel disease (IBD). Immune’s drug is currently in a Phase 2 trial targeting patients with moderate-to-severe ulcerative colitis (UC). Investors can view that article here >> LINK
I spent a lot of time looking at the IBD market for that article. Much of the background work I pulled from the Crohn’s & Colitis Foundation of America (CCFA) website; and as usual, I spent a day on PubMed just reading articles and abstracts on the IBD treatment paradigm and new therapeutic options under development. The clear competition for Immune is the approved biologic drugs, namely the tumor necrosis factor-alpha (TNF-α) drugs, infliximab (Remicade®, JNJ), adalimumab (Humira®, AbbVie), certolizumab pegol (Cimzia®, UCB), and golimumab (Simponi®, JNJ). In addition to these drugs, other approved biologics such as natalizumab (Tysabri®, Biogen) and vedolizumab (Entyvio®, Takeda) are commonly used in severe patients as well, and some very large players are in late-stage trials with drug that look to be generating solid results, including ozanimod and mongersen (Celgene), tofacitinib (Pfizer), etrolizumab (Roche), and ustekinumab (J&J).
One company, however, stuck out from the crowded “big pharma / biologic” strategy with a novel concept – a triple antibiotic in a single pill that targets what many researchers believe is the root cause of IBD, mycobacterium avium paratuberculosis (MAP). RedHill Biopharma (NASDAQ:RDHL) is currently in Phase 3 trials with RHB-104, a patent-protected combination of clarithromycin, clofazimine, and rifabutin in a single oral pill. I became intrigued with the concept of RHB-104 and thus decided to focus this article on my findings.
Quick Introduction to RedHill Biopharma (RDHL)
The company is well-funded, with $66 million in cash on the books as of July 2015 and no debt. The company is focused on late-stage clinical development of oral small molecules for the treatment of gastrointestinal and inflammatory conditions. RedHill has three ongoing Phase 3 programs in GI indications, including RHB-104 for Crohn’s disease (the subject of this article), RHB-105 for H. pylori infection, and Bekinda™ for gastroenteritis/gastritis and irritable bowel syndrome/diarrhea. Another late-stage asset, RHB-106, an oral capsule for bowel preparation ahead of colonoscopy or surgery, has been licensed to Salix Pharmaceuticals (now Valeant Pharma), which validates the company’s in-license/out-license strategy.
RedHill has several mid-stage candidates as well, including three oncology/inflammation candidates, Yeliva™, Mesupron®, and RP101, and a number of follow-on programs for the aforementioned late-stage drugs in additional indications. In addition, a marketing application has been filed for Rizaport™, a migraine drug in Europe (under review) and the U.S. (complete response). A snap-shot of the full pipeline can be seen below.
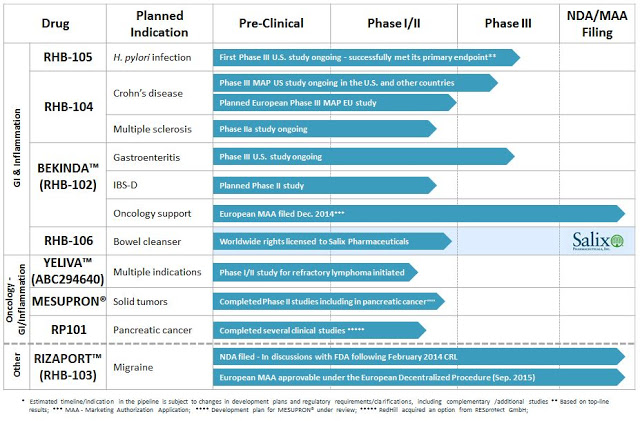
I like the focus of the company, which has aspirations of vertical integration in the U.S., the fact that they are well capitalized, led by what looks to be quality management, non-promotional in their dealings with the market, and backed by an impressive group of institutional investors (e.g. Orbimed, Broadfin, Special Situations, Visium, Longwood, Sabby, Rosalind, Fred Alger, etc…). The market capitalization is only $155 million and there are multiple shots on goal. Based on what I discovered in my brief look at the rest of the pipeline, I plan on doing several more articles on this interesting biopharma name. For now, below is my assessment of RHB-104 in Crohn’s disease.
A Quick Look At IBD
Inflammatory Bowel Disease (IBD), which includes ulcerative colitis and Crohn’s disease, is a chronic, lifelong condition with serious quality of life implications. According to the Crohn’s & Colitis Foundation of America (CCFA), IBD affects as many as 1.4 million Americans, most of which are diagnosed before the age of 30. Crohn’s disease accounts for approximately 65% of the IBD market. The primary characteristic of the disease is inflammation of the gastrointestinal (GI) tract, which manifests in symptoms that include persistent diarrhea, abdominal pain, cramping, rectal bleeding, and fatigue. Crohn’s disease is distinguished from ulcerative colitis in that any part of the GI tract can be affected by the inflammation, although the most commonly affected area is the small intestine (the ileum) near the ascending colon. Crohn’s disease may also appear in patches, affecting some areas of the GI tract while leaving other sections completely untouched. Crohn’s disease tends to manifest with more severe inflammation than UC and the ulcerations may extend through all layers of the bowel creating transmural perforations and strictures.
The cause of IBD is not entirely understood, but is believed to involve a number of factors including environmental influences, mucosal immune dysfunction, and genetic predisposition. It is only just recently that the concept of bacterial infection, specifically MAP, as a precursor and underlying cause for CD has gained acceptance. Nevertheless, there are no approved antibiotic therapies in the U.S. for CD.
Symptoms of IBD vary from person to person, and may change over time. With both Crohn’s disease and ulcerative colitis, patients go through periods of being symptom-free (remission) alternating with periods of having active disease symptoms (flare ups). For CD, the CCFA estimates patients are in remission or have mild disease activity approximately 50% of the time. Approximately 35% of CD patients will report moderate disease activity with at least one or two flare-ups during the course of a year. Approximately 10-15% will report severe and/or chronically active disease (Langholz E. et al., 1994).
The primary concern of moderate-to-severe disease is inflammation that leads to ulceration and perforation of the bowel wall leading to fistula (tunnel formations) to another part of the intestine, other internal organs, or to the skin surface. Left untreated, ulcers can lead to strictures (scar tissue) or abscesses that block the intestines and cause intense pain and discomfort. Patients with severe CD may also have nutritional deficiencies resulting from the inability to absorb proteins, vitamins, and fats through the damaged intestinal wall.
About 70% of people with CD eventually require surgery. Different types of surgical procedures may be performed depending on the reason for surgery, severity of illness, and location of the disease in the intestines. Unlike ulcerative colitis, surgery does not cure Crohn’s disease. Approximately 30% of patients who have surgery for CD experience recurrence of their symptoms within three years and up to 60% will have recurrence within ten years. (Sachar DB, 1990).
What We Know
In the decades since UC and CD were first identified, significant scientific progress has been made in understanding these chronic inflammatory diseases, particularly with respect to immunology, genetics, and microbiology. An increasing number of susceptibility genes have been identified, as have environmental and external factors including smoking and diet. The primary method for treating IBD is by knocking-down the body’s inflammatory response through the use of aminosalicylates, corticosteroids, and immunomodulators. When these drugs fail, and they typically do over 50% of the time (source: GlobalData), gastroenterologists turn to the more powerful biologic drugs noted above, with J&J’s Remicade® (infliximab) as the market leader.
According to J&J’s Phase 3 clinical trial with infliximab, 58% of moderate-to-severe CD patients responded to a single infusion of 5 mg/kg drug after two weeks. Unfortunately, the percent drops to only 18% placebo-adjusted after 30 weeks for the same dose used as a maintenance therapy (Hanauer SB, et al., 2002), and the best idea big pharma can come up with since infliximab approval is to simply double the dose and take more drug (Dumitrescu G, et al., 2015).
The lack of consistent and durable response to the TNF-α and other biologic drugs, along with the potentially dangerous side effects and risk of both infection and malignancies that can occur with long-term use, has led to some calling into question the pharmaco-economic benefit of these drugs. A study conducted by researchers at the Department of Public Health & Epidemiology University of Birmingham, UK concluded the anti-TNF drugs do offer a beneficial effect compared to standard-of-care, but that neither infliximab nor adalimumab is cost-effective as a maintenance therapy (Dretzke J, et al., 2011).
The MAP-Hypothesis
Drugs like the prednisolone, methotrexate, and the TNF-α and other biologic drugs treat only the symptoms of CD. To truly advance the treatment paradigm forward, researchers must start looking at the underlying causes of the disease. In this regard, an important link has been drawn between CD and another chronic inflammatory disease of the intestine that affects many animal species, including primates, called Johne’s disease (JD). Johne’s disease (also called paratuberculosis) is a contagious, chronic, and sometimes fatal infection that primarily affects the small intestine of ruminants (plant eating mammals). The disease was first discovered and characterized by Heinrich Albert Johne in 1895 and is unequivocally caused by mycobacterium avium paratuberculosis (MAP) (Wikipedia).
MAP is a slow growing obligate pathogenic bacterium, highly resistance to treatments with acid and alcoholic compounds due to a strong cellular wall with a high lipid composition. MAP is recognized as a multi-host mycobacterial pathogen with a proven specific ability to initiate and maintain systemic infection and chronic inflammation of the intestine of a range of histopathological types in many animal species, including primates. A 2007 survey conducted by the U.S. National Academy of Science Board of Agriculture found that nearly 70% of U.S. dairy herds are infected with MAP.
Significant similarities exist between Johne’s disease and Crohn’s disease, and scientists have spent the past decade characterizing the common neural and immune pathogenicities (Scanu AM, et al., 2007). Independent work conducted by a number of institutions has confirmed the link between CD and MAP infection (e.g. Hermon-Taylor, 2009, The Myble, 2013), with data out of the American Society of Microbiology finding MAP infection rates in patients with active and severe CD to be seven-times that of the normal healthy population. Another paper by Medoza JL, et al., 2010 found MAP DNA in 100% of recruited patients for a small study conducted in Spain. The authors found no MAP DNA in healthy controls, concluding that MAP infection might have clinical implications in Crohn’s disease. Quite interestingly, research shows that infliximab has antibiotic effects that reduce the level of MAP infection in patients with active and severe CD (Nazareth N, et al., 2015).
The “MAP-hypothesis” has gained enough steam that both the American Academy of Microbiology and the European Directorate General of Health and Consumer Protection conducted a separate, independent analysis of the link between MAP infection and incidence of CD.
The 2007 76-page EU-MAP Report makes the following conclusions:
Crohn’s Disease is most likely a multifactorial condition… The detection of MAP in a greater proportion of Crohn’s disease patients than in controls suggests that the organism may have a role either as a causative agent, as a secondary invader which exacerbates the disease or as a non pathogenic coloniser because of changed bowel conditions… If MAP is involved in the causation of Crohn’s disease, it would also require the presence of other susceptibility factors. It is also possible that its involvement may relate to a subset of Crohn’s disease cases… Results from recent drug trials in humans with drugs likely to be active against MAP have been encouraging but inconclusive. Larger scale double blind studies are in progress to obtain better data…
The 2008 41-page US-MAP Report makes the following conclusions:
Researchers and clinicians agree that onset of CD requires a series of events; implicated are certain inherited genetic traits, an environmental stimulus, and an overzealous immune and inflammatory response… Inflammation is normally caused by a “foreign body,” an inanimate object (i.e., splinter) or animate objects like rogue cells (i.e., cancer) or microorganisms (i.e., bacterium, virus, or fungus). Until the cause of inflammation is eliminated, the body continues to send in its clean-up crew, the white blood cells of inflammation whose job it is to expel the tissue invader. Inflammation only subsides when the causative agent is finally banished… There is suspicion, supported by reports of genetic inability to interact appropriately with certain bacteria or bacterial products in some patients, that CD may have a currently unrecognized infectious origin… One acknowledged potential microbial agent of CD is Mycobacterium avium subspecies paratuberculosis (MAP), a microorganism that causes a gastrointestinal disease similar to CD in ruminants, including dairy cattle, called Johne’s disease (or paratuberculosis). People with CD have 7:1 odds of having a documented presence of MAP in blood or gut tissues than those who do not have CD, thus the association of MAP and CD is no longer in question… Circumstantially, these observations appear to make a compelling case for MAP as involved in CD…
RHB-104: Proof-of-Concept in Crohn’s Disease
RHB-104 is a patented combination of three generic antibiotic ingredients, clarithromycin, rifabutin, and clofazimine, in a single oral capsule.- Clarithromycin is a broad-spectrum antibiotic with efficacy against Gram-positive infections such as Helicobacter pylori, Haemophilus influenzae, Streptococcus pneumoniae, and Streptococcus pyogenes. The drug was sold as Biaxin® by Abbott Labs but is now available as a generic in the U.S.
– Rifabutin is a semi-synthetic antibiotic with efficacy against both Gram-positive and Gram-negative bacteria, with primary use against mycobacteria for the treatment of tuberculosis.
– Clofazimine is a iminophenazine dye used in combination with other antibiotics for the treatment of leprosy and Mycobacterium avium infections in AIDS patients.
RHB-104 was developed to treat Mycobacterium avium paratuberculosis (MAP) infections. RedHill Biopharma acquired the drug from Giaconda Limited in August 2010. As part of the transaction, RedHill also acquired two additional drugs, RHB-105, a patented combination of omeprazole, amoxicillin, and rifabutin for the treatment of H. pylori bacterial infection in the gastrointestinal tract, and RHB-106, a patented formulation in tablet form intended for the preparation and cleansing of the gastrointestinal tract prior to abdominal procedures or colonoscopy.
Giaconda was founded in 2004 as a vehicle through which to commercialize therapies for gastrointestinal diseases and disorders developed by Professor Thomas Borody at the Centre for Digestive Diseases in Australia. Professor Borody licensed the patents to RHB-104 to RedHill Biopharma in 2010. Significant clinical work with RHB-104 or its components was conducted prior to the transaction with Gioconda.
In 1997, researchers out of George’s Hospital Medical School, London, UK tested rifabutin in combination with a clarithromycin or azithromycin. Results showed that of the 19 patients who were steroid dependent at the start of this study, only two continued to require steroids when treatment was established. A reduction in the Harvey-Bradshaw Crohn’s disease activity index occurred after 6 months treatment (P = 0.004) and was maintained at 24 months (P < 0.001) (Gui GP, et al., 1997). A separate study out of the Department of Medicine, University of Liverpool in the UK in 2000 showed an impressive response to clarithromycin in a group of patients with active Crohn’s disease, many of whom had been resistant to other therapy (Leiper K, et al., 2000).
The first human clinical work with the triple antibiotic combination was published by Professor Borody’s lab at the Center of Digestive Disease in Australia (Borody TJ, et al., 2002). The Phase 2a study in 12 patients with severe, obstructive or penetrating Crohn’s disease followed patients for a total of 54 months. Six out of 12 patients experienced a full response to the anti-MAP combination therapy achieving complete clinical, colonoscopic and histologic remission of Crohn’s disease. Four of these patients were able to cease treatment after 24-46 months, three of whom remained in total remission without treatment for up to 26 months and one patient relapsed after six months off treatment. A partial response was seen in two patients showing complete clinical remission with mild histologic inflammation. Return to normal of terminal ileal strictures occurred in five patients. This is as close to a cure for Crohn’s disease that researchers had seen to date.
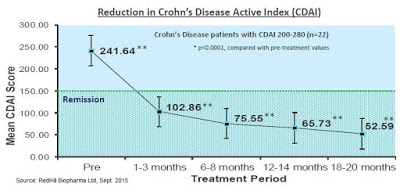
Professor Borody’s lab published additional results from a second study in 54 patients in 2005. Results showed that 60% of the patients achieved a significant reduction in Crohn’s Disease Activity Index (CDAI) of > 70 points, and of the total evaluable patients, 85% saw a decrease in CDAI from baseline, along with other measures of response such as mucosal healing. Patients also saw reductions in CD-related symptoms, including abdominal pain and diarrhea (Borody TJ, et al., 2005).
The figure below shows a 22 patient subset of the Borody study in which patients were stratified by disease severity. This more homogeneous group had 100% remission within less than 3 months. This is the strongest data seen to date in CD, and would essentially blow away competing late-stage data from mongersen – a drug that Celgene paid $710 million to acquire in 2014.
 Big Pharma Shows Up
Big Pharma Shows Up
Professor Borody’s lab licensed the rights to the triple antibiotic to Pharmacia for the Phase 3 program. Two hundred thirteen patients were randomized to 450 mg/day of rifabutin, 750 mg/day of clarithromycin, and 50 mg/day of clofazimine or placebo, in addition to a 16-week tapering course of prednisolone. Those in remission (Crohn’s Disease Activity Index ≤ 150) at week 16 continued their study medications in the maintenance phase of the trial. Primary endpoints were the proportion of patients experiencing at least one relapse at 12, 24, and 36 months.
At week 16, there were significantly more subjects in remission in the antibiotic arm (66%) than the placebo arm (50%; P = 0.02). Of 122 subjects entering the maintenance phase, 39% taking antibiotics experienced at least one relapse between weeks 16 and 52, compared with 56% taking placebo (P = 0.054). These results were encouraging, but the separation did not maintain statistical significance upon follow up at week 104, 26% vs. 43% relapse rate, respectively (P = 0.14) (Selby W, et al., 2007). The authors concluded the paper with the following statement:
Using combination antibiotic therapy with clarithromycin, rifabutin, and clofazimine for up to 2 years, we did not find evidence of a sustained benefit. This finding does not support a significant role for Mycobacterium avium subspecies paratuberculosis in the pathogenesis of Crohn’s disease in the majority of patients. Short-term improvement was seen when this combination was added to corticosteroids, most likely because of nonspecific antibacterial effects.
Following results of the Phase 3 study, Pfizer (acquired Pharmacia in 2003) decided not to continue development of the triple antibiotic for CD.
A Flawed Study
Following publication of the Phase 3 results by Pfizer, supporters of the triple antibiotic therapy everywhere called the study flawed. The initial criticism of the trial stemmed from the dosing strengths used, specifically with respect to under-dosing patients compared to the two studies conducted by Prof. Borody in the preceding years. The doses used in the 2005 Phase 2 study (n=54) were 600 mg/day of rifabutin, 1000 mg/day of clarithromycin, and 100 mg/day of clofazimine. Pfizer dosed 25% less rifabutin and clarithromycin and 50% less clofazimine in its trial. There were also serious dosing compliance issues with the Pharmacia Phase 3 trial.
A paper published by Gitlin L, et al., 2014 out of the Gastroenterology Institute, Las Vegas, Nevada cited the following criticism of the Phase 3 program run by Pfizer:
According to recommended dosing for Mycobacterium avium infection rifabutin was underdosed by >30%, clarithromycin by approximately 50%, and clofazimine by >50%—and then in the crucial part of the trial, as admitted by the authors—clofazimine did not dissolve properly. Clofazimine has a half-life of 70 days and would begin to be active about halfway through the 16-week induction of remission period. Once capsules stopped dissolving, its anti-DNA effect on the cell wall free mycobacteria was lost. Given the low doses and non-dissolution, it is highly likely that in the longer term the development of drug resistance—known to occur with mycobacteria—would have played a major role in the apparent increased relapse in the “active” group. According to Good Clinical Practice, 2 patients whose treatment violated the protocol should have been replaced by new trial patients where the clofazimine dissolved properly and was bioavailable. This raises the serious question of this trial’s validity beyond 16 weeks when the issue with clofazimine bioavailability commenced.
Several researchers/gastroenterologists had comments about the Pfizer data. J. Todd Kuenstner of the Memorial Hospital in West Allis, Wisconsin published this comment to the Selby paper:
The results of the Selby trial are encouraging (significantly better short-term remission rate in the treatment arm than the control arm) and suggest modifications of the current trial for future studies, which may lead to better outcomes… A blood culture method is now available to detect MAP infection in Crohn’s patients and could be used to assess MAP eradication in a properly designed controlled trial with more appropriate doses of the same drugs… The study design required an unrealistically high response rate for a positive outcome. Most of the current Crohn’s therapies including infliximab would probably fail to show a positive outcome by this measure… The study may not have had sufficient statistical power to show a more substantial benefit to a subset of the Crohn’s patients who might have benefited from the therapy. Studies show that approximately 50% of Crohn’s patients are infected with MAP… The other 50% may not benefit from therapy targeting MAP… In summary, the authors’ conclusions are not supported by their work.
Judith E. Lipton and David P. Barash of the University of Washington in Seattle, Washington published this comment in response to the Selby paper:
No attempt was made to culture or do polymerase chain reaction testing for MAP, either before or after treatment, so no conclusion can be drawn about the effect of their protocol on putative Mycobacterium avium paratuberculosis (MAP) infection. The MAP hypothesis of Crohn’s disease is not tested by this study, nor resolved in anything except the author’s assertions… Not only were the doses low, but the clofazimine was delivered in a double encapsulated form that may well have hampered bioavailability.
Professor Borody posted the comment below on the CDD website about Pfizer’s trial:
Unfortunately, the trial used sub therapeutic doses, failed to test patients before they were treated for MAP presence and did not replace patients who were given non dissolving Clofazimine drugs so that the long term maintenance part of the trial cannot be currently accepted as having any clinical significance.
In short, the general conclusion from peers is that the Pfizer study was vastly underpowered in terms of size, used insufficient doses of the key active ingredients, had major dosing issues with respect to drug bioavailability, never bothered to confirm MAP infection in participants, and looked at the wrong endpoint in relapse rates instead of remission in the ITT population.
RedHill Initiates A New Phase 3 Trial
In September 2013, RedHill initiated a new Phase 3 trial with RHB-104 in patients with moderate-to-severe active Crohn’s disease (NCT01951326). According to ClinicalTrials.gov, there are 83 sites in the U.S., Canada, Israel, Australia, New Zealand, and Europe enrolling patients, with a target total enrollment of 270 patients at 120 centers randomized 1:1 to RHB-104 vs. placebo. The primary endpoint of the study is the percent of patients in remission at week 26 (CDAI ≤ 100), with secondary endpoints including response rate at week 26, time to remission, duration of remission and response, and maintenance of remission from week 26 to 52. The daily doses of RHB-104 has been optimized to 450 mg rifabutin, 950 mg clarithromycin, and 100 mg of clofazimine. Data is expected late 2016.
Despite the fact that the Selby W, et al., 2007 concluded the triple antibiotic MAP therapy for CD holds no utility, the Phase 3 data actually provides very good support for RedHill achieving a positive outcome. For example, the primary endpoint for RedHill’s Phase 3 program is remission at 26 weeks. Looking at the Pfizer trial data, the under-dosed triple antibiotic achieved statistical significant at all time points tested on the remission endpoint.
A reanalysis of the Pfizer study was conducted by Behr & Hanley in 2008 and published in The Lancet. The authors took the data from the study and “reset” the baseline at week 16, looking at whether or not the 66% of the original patients in remission at week 16 maintained remission over the next two years. So instead of looking for relapse rates in the ITT population, the authors attempted to ascertain if patients that observed a benefit at week 16 (i.e. responders to anti-MAP therapy) saw persistent benefit at weeks 52 and 104. The results, presented below, show statistical significance at all time points.
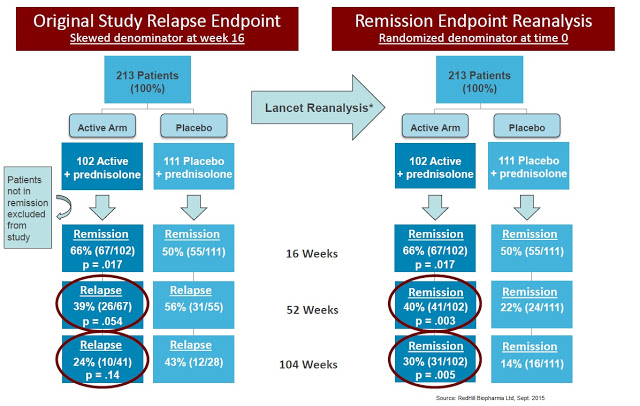
The data also compare remarkably well with J&J’s original Phase 3 study (ACCENT-1) with infliximab, which showed only a 39% remission rate at 30 weeks and 28% remission rate at 54 weeks. The data published by Selby W, et al., 2007 showed a 42% improvement in remission compared to infliximab responders and 2.5x the response rate on an ITT basis (40% vs. 16%, absolute). Bafflingly, Pfizer killed this program!
Pediatric Crohn’s – An Orphan Indication
In October 2013, RedHill presented a poster at the American College of Gastroenterology Annual Scientific Meeting showing positive results from retrospective study with 10 pediatric patients suffering from Crohn’s disease treated with RHB-104 by Professor Borody at the Centre for Digestive Diseases in Sydney, Australia. The result of the retrospective analysis demonstrated a clinical remission rate of 80% in 10 pediatric Crohn’s disease patients, aged 8 to 17 (median: 14.3), treated for six to 117 months. The state of remission was assessed and recorded regularly throughout treatment and was defined as Pediatric Crohn’s Disease Activity Index (PCDAI) < 10 for a minimum of three months, with stable, or reduction in, preexisting Crohn’s disease medications. RedHill obtained U.S. FDA Orphan Drug status for RHB-104 for the treatment of Crohn’s disease in the pediatric population.
An Enormous Market For RHB-104
RedHill Bio’s RHB-104 is patent protected to 2029, with potential expansion of that under the GAIN act to 2034. Yes, the components of RHB-104 are three generic molecules, but given the optimized dosing of the single capsule and the fact that compounding pharmacies cannot legally recreate the formulation under the FDA’s Drug Quality and Security Act of 2013, I believe RHB-104 has no significant IP issues. In addition, clofazimine, only approved for the treatment of leprosy, it scarcely available in the U.S. and EU, and unlikely to prescribed as part of any unapproved treatment regimen outside of U.S. FDA approval. Additionally, U.S. FDA Orphan Drug status for pediatric Crohn’s provides, at a minimum, seven years of market exclusivity for the company. Investors should know, in September 2015, the company entered into a manufacturing and supply agreement with Corealis Pharma for RHB-104.
According to the CCFA, there are an estimated 900,000 patients in the U.S. suffering from Crohn’s disease. There are easily another 1.1 million patients in commercially viable regions outside the U.S., such as Canada, Europe, and Israel. Approximately 50% of these 2.0 million patients are in remission or suffer from only mild disease. Another 750,000 or so patients suffer from moderate disease and may achieve adequate response to aminosalicylates, corticosteroids, and immunomodulators. This leaves approximately 250,000 CD patients worldwide that seek advanced treatment options such as the TNF-α and other biologics.
Using the Phase 3 infliximab data as a point of reference, 50-60% of patients ultimately fail on these biologic drugs, and 70% eventually require some sort of surgery resulting from chronic CD. The TNF-α drugs and biologics such as natalizumab are also dangerous, and carry FDA (“Black Box”) warnings for risk of infection (Nanau RM, et al., 2014) and hematologic malignancies. They are also incredible expensive drugs, ranging in price from $20,000 to over $65,000 per year based on dose.
I see an enormous opportunity for RHB-104, not only as a treatment option for patients failing biologic medications, but as a first-line therapy in patients with confirmed MAP infection as a potential cure for CD. In this regard, RedHill is developing a diagnostic test for MAP infection in collaboration with Quest Diagnostics. This should both help improve the efficacy outcome of RHB-104 and provide meaningful pharmacoeconomic benefit in the eyes of payors once the drug is approved. Plus, RHB-104 has demonstrated good safety and it is an oral medication. All the biologic medications are IV/infusion.
I think RHB-104 is easily a multi-hundred million dollar drug. Pricing remains a wildcard at this point, but I’m going to assume that RHB-104 costs $12,000 per year ($1,000 per month). This is about 65% cheaper than the biologic drugs and investors should keep in mind that this is still very cheap for an Orphan Drug. It is about what I suspect the pricing will be for biosimilar infliximab in the U.S., once approved. Using some conservative assumptions that 50% of severe CD patients have MAP infection and that 50% will fail biologics, the target population for RHB-104 under a refractory label is still 62,500 patients in the U.S.+EU markets. At 10% penetration, RHB-104 is a $500 million drug.
However, I believe RHB-104 has the potential to: 1) have penetration rates far above 10% in the MAP-confirmed refractory population, and 2) move ahead of the biologics based on the development of the companion diagnostic. This could ultimately increase the peak sales of RHB-104 far in excess of billion dollars on a global basis.
Final Thoughts On RHB-104
RedHill Bio’s RHB-104 looks like a very interesting candidate and a potential paradigm changer for the treatment of Crohn’s disease. It’s a novel idea with demonstrated proof-of-concept in both early-stage and late-stage human trials. It looks to be safe and is a convenient oral capsule. At one point, pharmaceutical giant Pharmacia was interested in the asset, and despite what are obvious areas for improvement with respect to the dosing and protocol design, some believed the trial to be a success. Dr. William M. Chamberlin of the Mountain View Regional Medical Center in Las Cruces, New Mexico said in 2007 following the publication of the data by Selby W, et al. that, “The anti-MAP therapy response rate is better than any other therapy, including infliximab, to date.” He called the drug a potential “miracle therapy” for Crohn’s patients.
RedHill obviously sees significant potential for RHB-104 in CD and is investing heavily in the current Phase 3 and additional human clinical trials. For example, the company is also testing RHB-104 in patients where MAP infection is hypothesized to play a role in disease pathophysiology. Interesting indications include multiple sclerosis, rheumatoid arthritis, psoriasis, and lupus. Management is investigating RHB-104 in a Phase 2a study in 18 patients with multiple sclerosis (NCT01717664). The trial is fully-enrolled and data is expected in the next few months. There are several available papers on PubMed suggesting a link between MS and MAP-infection, although this will have to be a topic for a separate article!
The opportunity for RedHill Biopharma with RHB-104 looks significant. If the Phase 3 trial is successful, I think the market will start to view RHB-104 as having peak sales in the $500-750 million range for the U.S., with potential expansion to over one billion on a global basis. The market capitalization is only $155 million, and the company has a strong and debt-free balance sheet with approximately $66 million in cash as of the end of July 2015. This article covers only RHB-104, and says nothing about the two other Phase 3 assets, RHB-105 and Bekinda (RHB-102), as well as RHB-106, currently in late-stage development at Valeant Pharmaceuticals and a host of mid-stage candidates for various oncology indications. For my next article on the company, I plan to tackle some of these additional candidates. I hope I find them to be as intriguing as I did RHB-104.
——
BioNap is party to a services agreement with the company that is the subject of this report pursuant to which BioNap is paid five thousand dollars per month by the company in exchange for the provision of research reports, investor relations services, and general consulting services. Please see additional information on our Disclaimer.


